New genetic-modification approach could eliminate sleeping sickness
 Researchers at the Oregon State University (OSU) have devised innovative approach that could reduce or even eliminate the deadly disease African trypanosomiasis (sleeping sickness), which was shown in a recent mathematical modeling study. They developed genetic-modification strategies to reduce the transmission of sleeping sickness, but this method appears as a promising strategy for reducing the incidence of others vector-borne diseases.
Researchers at the Oregon State University (OSU) have devised innovative approach that could reduce or even eliminate the deadly disease African trypanosomiasis (sleeping sickness), which was shown in a recent mathematical modeling study. They developed genetic-modification strategies to reduce the transmission of sleeping sickness, but this method appears as a promising strategy for reducing the incidence of others vector-borne diseases.
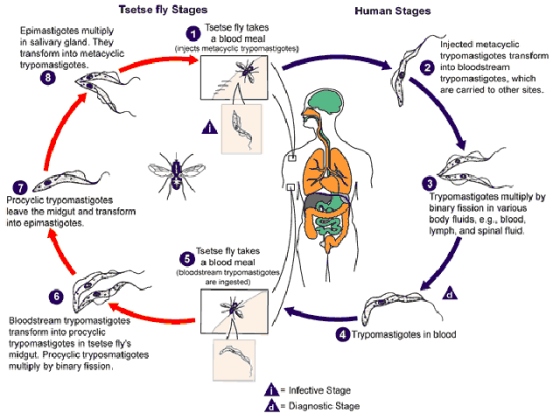
Sleeping sickness is a parasitic disease of humans and other animals, caused by protozoa and carried by the tsetse fly. It infects 30,000 people in sub-Saharan Africa each year and is almost always fatal without treatment. Sleeping sickness causes serious mental and physical deterioration, including the altered sleep patterns that give the disease its name.
Currently, control efforts primarily target the tsetse fly by insect traps, insecticide spraying of land and livestock, and sterile-insect technique. These strategies have been of some value, but only in limited areas and the effects aren’t permanent.
The research team genetically modified tsetse fly to be resistant to carrying the sleeping sickness parasite. They created a genetically modified version of the Sodalis bacteria, commensal bacteria that colonize the gut of a tsetse fly.
In order to drive the genetically modified tsetse into fly populations, researchers used bacteria called Wolbachia. These bacteria are transmitted by female insects to their progeny.
According to the researchers, when the GMO version of Sodalis bacteria is driven into fly populations, it would kill the disease-causing trypanosome parasite. Once this process begins it should spread widely and be self-sustaining, so it would not be necessary to constantly drive GMO flies in the huge geographic areas of Africa. The flies carrying the Wolbachia bacteria should naturally increase their populations and have an inherent survival advantage over conventional tsetse flies.
However, there are a few obstacles of this approach but the research shows that dealing with all of those obstacles should be possible. Some of them include: the transgenic Sodalis has to be reasonably effective (about 85 percent) at blocking the parasite; the Wolbachia bacteria, which has some effect on the health of flies affected with it, must not kill too many of them in order to spread those genes; and the target species of tsetse fly has to be a majority of the tsetse flies in the treated areas.
“The analysis is clear that this approach has significant promise of success. Because of the relatively low infectiousness of the parasite and the ability of Wolbachia to drive the resistance genes, no part of the system has to be 100 percent perfect in order to ultimately achieve near eradication of this disease”, said Jan Medlock, an assistant professor in the OSU Department of Biomedical Sciences.
Genetic-modification strategies could open new approaches to deal with other tropical, insect-carried diseases of even greater importance, including dengue fever and malaria. However, dealing with malaria is difficult, because that disease historically has shown a remarkable ability to mutate. On the other hand, the research team is confident that some short-term gains are possible with the new approach.
For more information, read the paper published in the journal PLOS Neglected Tropical Diseases: “Evaluating Paratransgenesis as a Potential Control Strategy for African Trypanosomiasis” [470KB PDF].









Magnificent job!