Silk brain implants may reduce the progression of epilepsy
 A group of researchers at Legacy Research Institute, Oregon Health and Sciences University (OHSU) and Tufts University have developed silk implants that they have placed in the brain of laboratory rats in order to reduce the progression of epilepsy. These silky brain implants are designed to release adenosine (ADO) – a specific chemical compound which has been shown to play an important part in fight against epilepsy.
A group of researchers at Legacy Research Institute, Oregon Health and Sciences University (OHSU) and Tufts University have developed silk implants that they have placed in the brain of laboratory rats in order to reduce the progression of epilepsy. These silky brain implants are designed to release adenosine (ADO) – a specific chemical compound which has been shown to play an important part in fight against epilepsy.
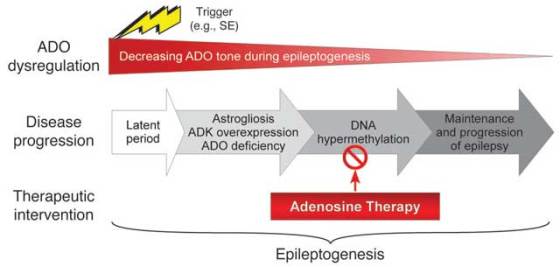
Epilepsy is currently the third most common neurological disorder that affects nearly 50 million people worldwide. It is connected with recurring seizures that tend to become more frequent and severe over time. It has been shown that ADO decreases neuronal excitability and helps stop seizures. Also, previous studies suggested that abnormally low levels of ADO may be linked to epilepsy.
The research team investigated long-term effects of an ADO-releasing silk-implant therapy in rats. They claim that ADO’s beneficial effects are due to epigenetic modifications, which alter gene expression without modifying the underlying DNA sequence. Example of such modifications is DNA methylation – a process which involves the addition of a methyl group to the DNA nucleotides affecting which genes are accessible and can be turned on. Genes are more likely to turn on when methyl groups have been taken away (demethylated).
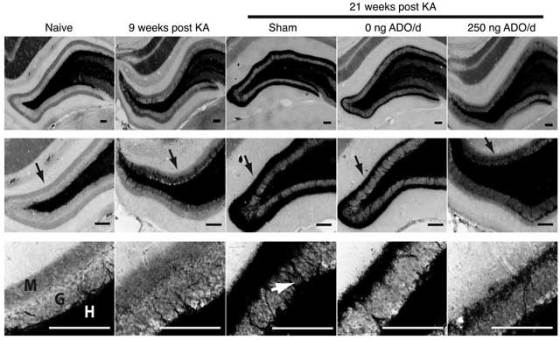
Researchers demonstrated that changing ADO levels affects DNA methylation in the brain. Lower levels of DNA methylation were associated with greater amounts of ADO. They also found that rats induced to develop epilepsy have higher levels of methylated DNA. Moreover, epileptic rat brains that had been treated with the ADO-releasing silk implant exhibited DNA methylation levels similar to brains of normal rats. This therapy also significantly reduced the worsening of the epilepsy over time.
The researchers have also found that increase in mossy fiber sprouting, the formation of new excitatory circuits in the part of the brain where seizures usually originate, is one mechanism involved in a specific type of the epilepsy. Animals that received the implants showed less sprouting than animals that were not given the drug.
“Based on our findings that 10 days of adenosine delivery prevented the sprouting of mossy fibers for at least three months in rats, we predict that the benefits of our adenosine therapy may extend even longer. However, this assumption needs to be validated in long-term experiments that go beyond three months”, said Detlev Boison, Director of Neurobiology Research at Legacy Research Institute and Adjunct Professor at OHSU.
Many previous studies on anti-epileptic drugs often tested the treatments too early. In this study, the rats received the implants only after they experienced a number of seizures. According to the researchers, when the therapy is applied too early, it interferes with the trigger for epilepsy and it is less severe. However, this doesn’t necessarily stop the progression of the decease. They showed that ADO-releasing silk did not completely abolish seizures in their rats but reduced them four-fold.
ADO-releasing silk brain implants are biodegradable, safe to use in rats and one day they may be used in the clinic. The release of ADO occurs for 10 days and then the silk completely dissolves.
“Clinical applications could be the prevention of epilepsy following head trauma or the prevention of seizures that often — in about 50 percent of patients — follow conventional epilepsy surgery. In this case, adenosine-releasing silk might be placed into the resection cavity in order to prevent future seizures”, said Boison.
Future studies will need to determine silk implants optimal use and safety for humans before they become used in medical applications. Supported by National Institute of Neurological Disorders and Stroke (NINDS), Legacy Health Institute researchers need to examine the efficacy of different doses of ADO, the duration of ADO release as well as how long the effects of the ADO-releasing silk implant will last.
For more information, read the article published in the Journal of Clinical Investigation: “Epigenetic changes induced by adenosine augmentation therapy prevent epileptogenesis” [2.4MB PDF].









Biomimicry at its best.
Dr.A.Jagadeesh Nellore(AP),India