VisionCare’s Implantable Miniature Telescope approved by FDA
 VisionCare Ophthalmic Technologies, Inc., a developer of advanced visual prosthetic devices, announced that the U.S. Food and Drug Administration (FDA) approved the company’s Implantable Miniature Telescope which is used to improve vision in patients with end-stage age-related macular degeneration (AMD). VisionCare’s telescope implant is integral to a new patient care program, CentraSight, for treating patients with end-stage macular degeneration.
VisionCare Ophthalmic Technologies, Inc., a developer of advanced visual prosthetic devices, announced that the U.S. Food and Drug Administration (FDA) approved the company’s Implantable Miniature Telescope which is used to improve vision in patients with end-stage age-related macular degeneration (AMD). VisionCare’s telescope implant is integral to a new patient care program, CentraSight, for treating patients with end-stage macular degeneration.
“The telescope implant represents a new category of treatment for this severely visually impaired population,” said Allen W. Hill, CEO of VisionCare. “This approval is the culmination of years of scientific and clinical development. We are excited to now provide this new technology and related CentraSight treatment program to the ophthalmic community to help their patients with this devastating disease gain improved vision and quality of life. This day would not be possible without the steadfast commitment of our clinical investigators, employees, and venture capital investors.”
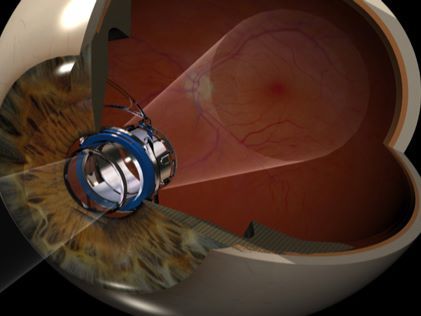
The Implantable Miniature Telescope, work of Dr. Isaac Lipshitz, is indicated for monocular implantation to improve vision in patients greater than or equal to 75 years of age with stable severe to profound vision impairment (best-corrected distance visual acuity 20/160 to 20/800) caused by bilateral central scotomas (blind areas) associated with end-stage AMD. This level of visual impairment constitutes statutory (legal) blindness.
“This is truly a breakthrough technology for AMD patients as their treatment options have been limited until now,” said Kathryn A. Colby, M.D., Ph.D., ophthalmic surgeon at Massachusetts Eye and Ear Infirmary in Boston and an Assistant Professor of Ophthalmology at Harvard Medical School. “The clinical results from the pivotal FDA trial have proven we can place this tiny telescope prosthesis inside the eye to help patients see better and, for some, even to levels at which they can recognize people and facial expressions that they could not before.”
The magnification provided by the implant reduces the impact of the blind spot caused by end-stage AMD. Endstage AMD causes severe to profound central vision loss in both eyes due to either wet AMD that has progressed to scarring of the macula despite drug treatments, or dry AMD that has progressed to geographic atrophy, the most advanced form of dry AMD.
Smaller than a pea (3.6 mm diameter; 4.4 mm length), the telescope is implanted in one eye in a surgical procedure. In the implanted eye, the device renders enlarged central vision images over a wide area of the retina to improve central vision, while the non-operated eye provides peripheral vision for mobility and orientation.
Results from the two U.S. clinical trials, conducted at 28 leading ophthalmic centers, showed that patients achieved clinically meaningful gains in visual acuity and quality of life with the telescope implant.
“The published outcomes from these rigorous trials attest to the robust and sustained benefits we were able to attain with the telescope implant for this underserved population,” remarked Stephen S. Lane, M.D., Adjunct Professor of Ophthalmology, University of Minnesota, in private practice at Associated Eye Care, Stillwater, MN, and the Medical Monitor for the telescope implant clinical trials.
VisionCare will conduct a post-approval study to monitor patient outcomes under commercial conditions and a second smaller study will follow clinical trial patients for an additional two years. The risks and benefits associated with the telescope implant are discussed in the Patient Information Booklet available at CentraSight webpage.









Hi. I have a problem with my eye and it is diagnosed as ‘bilateral optic atrophy macular degeneration’. My doctor give me a report about my illness that says I need 3x long and 2x short telescope optic. I want to know that is one of your products suitable for me? If your answer is yes how can I get it? I am writting you from Turkey. Is there any place in Turkey in relation with you? If ı will not satisfy with your product, is giving it back possible? and the last question, how much will it take approximately? l want to buying miniature telescope. Can you help me plaese Best regards…