Major breakthrough for iron-air batteries
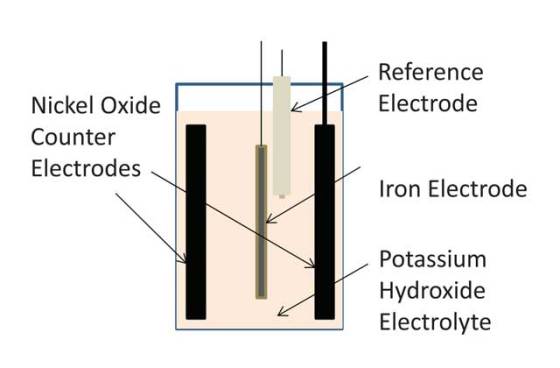
 A team of researchers from University of Southern California (USC) has developed an air-breathing battery that uses the chemical energy generated by the oxidation of iron plates that are exposed to the oxygen in the air. The technology relies on a process similar a process similar to rusting, and it could lead to new generation of affordable, rechargeable and eco-friendly batteries.
A team of researchers from University of Southern California (USC) has developed an air-breathing battery that uses the chemical energy generated by the oxidation of iron plates that are exposed to the oxygen in the air. The technology relies on a process similar a process similar to rusting, and it could lead to new generation of affordable, rechargeable and eco-friendly batteries.
Iron-air batteries saw a surge in interest during the 1970s energy crisis, but a competing chemical reaction of hydrogen generation that takes place inside the battery (known as hydrolysis) wasted about 50 percent of the battery’s energy, making it too inefficient to be useful. Led by Sri Narayan, professor of chemistry at the USC Dornsife College of Letters, Arts and Sciences, the researchers managed to reduce the energy loss down to 4 percent, making iron-air batteries that are about 10 times more efficient than their predecessors.
The team managed to achieve this by adding a very small amount of bismuth sulfide into the battery. Bismuth, which happens to be part of the active ingredient in Pepto-Bismol and helps give the pink remedy its name, shuts down the wasteful hydrogen generation. We achieve a ten – fold reduction in hydrogen evolution
“Adding lead or mercury might also have worked to improve the battery’s efficiency but wouldn’t have been as safe”, said Narayan. “A very small amount of bismuth sulfide doesn’t compromise on the promise of an eco-friendly battery that we started with.”
The team developed batteries with the capacity to store between eight and 24 hours’ worth of energy, a high charging-efficiency of 96%, a high discharge capacity of 0.3 Ah/g, and also a twenty-fold increase in capacity for the two-hour discharge rate. Tests performed by the researchers shown that the efficiency and discharge performance were stable with repeated cycling.
I would like to point out that despite the fact the researchers managed to nearly double its effectiveness compared to past models, iron-air batteries are known for having the lowest energy density (Wh/kg) compared to other types of metal-air batteries. Compared to aluminum-air batteries it has roughly 3 times lower energy density, and lithium-air batteries have roughly 11 times more energy density compared to iron-air batteries.
Although not suitable for use in electric vehicles or lightweight gadgets, these batteries could be used to store energy generated by solar or wind power. Batteries have typically not been a viable solution for utilities, and rechargeable lithium-ion batteries are at least 10 times as expensive as iron-air batteries. In order to improve the technology, USC researchers are looking for ways to increase the battery storage with less material used.
For more information, you can read the article published in the Journal of the Electrochemical Society: “A High-Performance Rechargeable Iron Electrode for Large-Scale Battery-Based Energy Storage” [0.98MB PDF].









Innovative.
Dr.A.Jagadeesh Nellore(AP),India
E-mail: anumakonda.jagadeesh@gmail.com
Spot on with the remark about other metal-air batteries and the potential of iron-air batteries.