Research led to faster charging batteries capable to retain capacity
 A group of researchers from University of Illinois developed a three-dimensional nanostructure for battery cathodes that allows dramatically faster charging and discharging without sacrificing energy storage capacity. Aside from quick-charge consumer electronics, batteries that can store a lot of energy, release it fast and recharge quickly are desirable for electric vehicles, medical devices, lasers and military applications.
A group of researchers from University of Illinois developed a three-dimensional nanostructure for battery cathodes that allows dramatically faster charging and discharging without sacrificing energy storage capacity. Aside from quick-charge consumer electronics, batteries that can store a lot of energy, release it fast and recharge quickly are desirable for electric vehicles, medical devices, lasers and military applications.
“This system that we have gives you capacitor-like power with battery-like energy”, said lead researcher Paul Braun, a professor of materials science and engineering at Illinois. “Most capacitors store very little energy. They can release it very fast, but they can’t hold much. Most batteries store a reasonably large amount of energy, but they can’t provide or receive energy rapidly. This does both.”
Making the active material in the battery as a thin film allows it to charge and discharge much faster, however, it also significantly reduces the capacity because the active material lacks volume to store energy. Braun’s group counters that by wrapping a thin film into a three-dimensional structure, achieving both high active volume (high capacity) and large current (fast charge and discharge). All of the processes the group used are also used at large scales in industry so the technique could be scaled up for manufacturing.
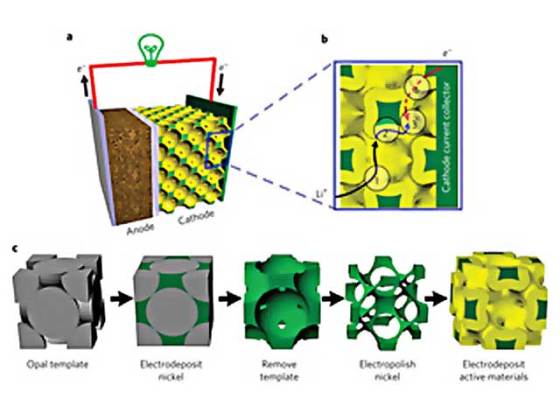
The key to the group’s novel 3-D structure is self-assembly. They begin by coating a surface with tiny spheres, packing them tightly together to form a lattice. Trying to create such a uniform lattice by other means is time-consuming and impractical, but the inexpensive spheres settle into place automatically.
Then the researchers fill the space between and around the spheres with metal. The spheres are melted or dissolved, leaving a porous 3-D metal scaffolding, like a sponge. Then a process called electropolishing uniformly etches away the surface of the scaffold to enlarge the pores and make an open framework. Finally, the researchers coat the frame with a thin film of the active material.
The result is a bicontinuous electrode structure with small interconnects, so the lithium ions can move rapidly; a thin-film active material, so the diffusion kinetics are rapid; and a metal framework with good electrical conductivity.
They have demonstrated battery electrodes that can charge or discharge in a few seconds, 10 to 100 times faster than equivalent bulk electrodes, yet can perform normally in existing devices. The group demonstrated both NiMH and Li-ion batteries, but the structure is general, so any battery material that can be deposited on the metal frame could be used.
“We like that it’s very universal, so if someone comes up with a better battery chemistry, this concept applies”, said Braun, who is also affiliated with the Materials Research Laboratory and the Beckman Institute for Advanced Science and Technology at Illinois. “This is not linked to one very specific kind of battery, but rather it’s a new paradigm in thinking about a battery in three dimensions for enhancing properties.”
This kind of performance could lead to phones that charge in seconds or laptops that charge in minutes, as well as high-power lasers and defibrillators that don’t need time to power up before or between pulses. Braun is particularly optimistic for the batteries’ potential in electric vehicles, because battery life and recharging time currently present major limitations of electric vehicles.
For more information, you can read the paper published in Nature Nanotechnology named: “Three-Dimensional Bicontinuous Ultrafast-Charge and -Discharge Bulk Battery Electrodes”.









Excellent. This will help Battery charging by Renewables like Solar/Wind.
Dr.A.Jagadeesh Nellore(AP),India