Stanford scientists develop high-efficiency zinc-air battery
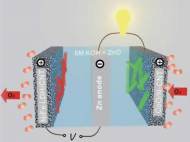 While lithium-ion batteries present the most dominant rechargeable battery on the market, their limited energy density (energy stored per unit volume), high cost and safety problems drives various research groups to invent a battery with better performance. Stanford University researchers have developed an advanced zinc-air battery which could serve low-cost alternative to conventional lithium-ion batteries.
While lithium-ion batteries present the most dominant rechargeable battery on the market, their limited energy density (energy stored per unit volume), high cost and safety problems drives various research groups to invent a battery with better performance. Stanford University researchers have developed an advanced zinc-air battery which could serve low-cost alternative to conventional lithium-ion batteries.
“There have been increasing demands for high-performance, inexpensive and safe batteries for portable electronics, electric vehicles and other energy storage applications”, said Hongjie Dai, a professor chemistry at Stanford and lead author of the study. “Zinc-air batteries are attractive because of the abundance and low cost of zinc metal, as well as the non-flammable nature of aqueous electrolytes, which make the batteries inherently safe to operate.”
Since these batteries rely on oxygen from the atmosphere, metal-air batteries have drastically higher theoretical energy density than either traditional aqueous batteries or lithium-ion batteries. Zinc-air batteries combine atmospheric oxygen and zinc metal in a liquid alkaline electrolyte to generate electricity with a byproduct of zinc oxide. When the process is reversed during recharging, oxygen and zinc metal are regenerated.
“Primary (non-rechargeable) zinc-air batteries have been commercialized for medical and telecommunication applications with limited power density. However, it remains a grand challenge to develop electrically rechargeable batteries, with the stumbling blocks being the lack of efficient and robust air catalysts, as well as the limited cycle life of the zinc electrodes”, said Dai.
Active and durable electrocatalysts on the air electrode are required to catalyze the oxygen-reduction reaction during discharge and the oxygen-evolution reaction during recharge. However, both catalytic reactions in most known zinc-air batteries are sluggish. Recently, a group of researchers at Dai’s research lab developed a number of high-performance electrocatalysts made with non-precious metal oxide or nanocrystals hybridized with carbon nanotubes.
“We found that similar catalysts greatly boosted the performance of zinc-air batteries”, said Dai. “A combination of a cobalt-oxide hybrid air catalyst for oxygen reduction and a nickel-iron hydroxide hybrid air catalyst for oxygen evolution resulted in a record high-energy efficiency for a zinc-air battery, with a high specific energy density more than twice that of lithium-ion technology.”
These catalysts produced higher catalytic activity and durability in alkaline electrolytes than catalysts made with platinum and other precious metals. The novel battery also demonstrated good reversibility and stability over long charge and discharge cycles over several weeks.
“This work could be an important step toward developing practical rechargeable zinc-air batteries, even though other challenges relating to the zinc electrode and electrolyte remain to be solved”, said Dai.
For more information, read the paper published in the journal Nature Communications: “Advanced zinc-air batteries based on high-performance hybrid electrocatalysts”.









Leave your response!