Using phthaloyl peroxide to streamline the manufacture of phenols
 Researchers at the University of Texas at Austin and University of California, Los Angeles, have discovered a new chemical reaction that has the potential to lower the cost and streamline the manufacture of compounds ranging from agricultural chemicals to pharmaceutical drugs. The reaction resolves a long-standing challenge in organic chemistry in creating phenolic compounds from aromatic hydrocarbons in a fast and low-cost manner.
Researchers at the University of Texas at Austin and University of California, Los Angeles, have discovered a new chemical reaction that has the potential to lower the cost and streamline the manufacture of compounds ranging from agricultural chemicals to pharmaceutical drugs. The reaction resolves a long-standing challenge in organic chemistry in creating phenolic compounds from aromatic hydrocarbons in a fast and low-cost manner.
Phenolic compounds, or phenols, are broadly used as disinfectants and fungicides, as well as in medicine in both traditional ways when used form a natural source and drugs that are being tested when phenols are synthetically created. Synthetic creation of phenol seems deceptively simple – all it requires is replacing a hydrogen molecule on an aromatic hydrocarbon with an oxygen molecule.
“This is a chemical transformation that is underdeveloped and at the same time pivotal in the production of many chemicals important to life as we know it”, said Dionicio Siegel, an assistant professor of chemistry in the College of Natural Sciences at The University of Texas at Austin.
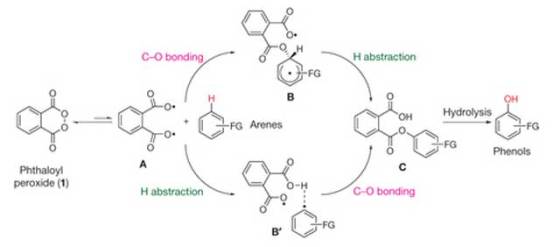
Siegel and his colleagues decided to try to simplify the transformation process where aromatic hydrocarbons are turned into phenols by using phthaloyl peroxide – a chemical studied in the late 1950s and early 1960s, but it has been largely ignored during the intervening years. The advantage to using phthaloyl peroxide is that the reaction does not require the use of acids or catalysts to work, and it can add oxygen to a wide variety of starting materials.
According to the researchers, there are no special conditions or complicated steps within the process. After combining and mixing of aromatic hydrocarbons and phthaloyl peroxide, you end up with phenols.
The new process can be applied to other problems in organic chemistry. One particular area of interest is creating metabolites to drugs. Metabolites are the products left after the body finishes breaking down, or metabolizing, a substance. When testing drugs, scientists need to take into account not just how the drug itself reacts in the body, but also how the metabolites react.
“We’ve had a long-standing interest in accessing metabolites of drugs or compounds that are used in biological systems”, said Siegel. “Just as it’s important that the drug doesn’t have deleterious side effects, it’s equally important that the metabolite doesn’t have an effect. You need to be able to test them, and there’s not really a direct way to access metabolites other than using liver microsomes, and that’s not efficient and it doesn’t always work.”
Another area where University of Texas at Austin researchers and their colleagues are applying what they have learned is in developing even more reactive agents that will expand the scope of chemicals that can be transformed. Siegel hopes to get this new process into the hands of others research groups quickly, and he is working with chemical supply companies to increase the availability of the phthaloyl peroxide compound as well as the precursors needed to create it.
For more information, read the article published in Nature: “Metal-free oxidation of aromatic carbon–hydrogen bonds through a reverse-rebound mechanism”.
Make sure to check the supplementary information paper [12.2MB PDF], where you can find 141 pages of data related to this research.










Leave your response!